AVAILABILITY AND STORAGE OF VACCINES IN COMMUNITY PHARMACIES IN DELTA STATE
INTRODUCTION
Immunization is the process by which an individual’s immune system becomes fortified against an agent (known as the immunogen). When this system is exposed to molecules that are foreign to the body, called non-self, it will orchestrate an immune response, and it will also develop the ability to quickly respond to a subsequent encounter because of immunological memory. This is a function of the adaptive immune system. Therefore, by exposing an animal to an immunogen in a controlled way, its body can learn to protect itself; this is called active immunization (Okwor, et al., 2012)
The most important elements of the immune system that are improved by immunization are the T cells, B cells, and the antibodies B cells produce. Memory B cells and memory T cells are responsible for a swift response to a second encounter with a foreign molecule. Passive immunization is direct introduction of these elements into the body, instead of production of these elements by the body itself.
Immunization is done through various techniques, most commonly vaccination. Vaccines against microorganisms that cause diseases can prepare the body’s immune system, thus helping to fight or prevent an infection. The fact that mutations can cause cancercells to produce proteins or other molecules that are known to the body forms the theoretical basis for therapeutic cancer vaccines.
Other molecules can be used for immunization as well, for example in experimental vaccines against nicotine (NicVAX) or the hormone ghrelin in experiments to create an obesity vaccine. Immunizations are definitely less risky and an easier way to become immune to a particular disease by risking a milder form of the disease itself. They are important for both adults and children in that they can protect us from the many diseases out there.
Through the use of immunizations, some infections and diseases have almost completely been eradicated throughout the United States and the World. One example is polio. Thanks to dedicated health care professionals and the parents of children who vaccinated on schedule, polio has been eliminated in the U.S. since 1979 (American Pharmaceutical Association [Apha], 2013).
Polio is still found in other parts of the world so certain people could still be at risk of getting it. This includes those people who have never had the vaccine, those who didn’t receive all doses of the vaccine, or those traveling to areas of the world where polio is still prevalent… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
Problem Statement
More than 40,000 to 50,000 adult and child death could have been prevented annually in Nigeria if there was a successful routine immunization for certain preventable diseases of which include measles, herpes zoster, tetanus and a host of others (Abdhuraheem, et al., 2011).
The federal government and donor agencies make so much effort and spend close to 50 billion dollars annually in the supply chain of vaccines but when these monies are spent and the purpose for which they are spent are not achieved due to a reduced potency of such vaccines or due to inadequate manpower for vaccine delivery to the target population. It can be said to be an investment in futility… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
Research Objectives
The objective of this research is to access the availability and storage of vaccines in community Pharmacies in Delta state.
- Specific Objectives
The specific objectives of this study are outlined as follows; (Scroll down for the link to get the Complete Chapter One to Five Project Material)

LITERATURE REVIEW
This chapter reviews various research studies and literature on the effects and consequences brought about by improper vaccine availability systems and poor vaccine storage and handling to the community. The chapter examines various research studies and reports done locally (Nigeria), regionally (Africa), and globally. The chapter also reviews some common types of vaccines currently available in routine immunization at a healthcare facility for humans.
Global perspective on vaccines availability and management
The study conducted in Bali province Indonesia regarding improving the animal health cold chain and vaccine management indicated that there were urgent needs for improvements in the management of vaccines. Approximately half of the refrigerators were unsuitable for vaccine storage generally in poor condition, the temperature was not monitored.
As a result healthcare workers did not know if the temperature of the refrigerator was within the recommended range at 2-8 0C. In addition, vaccines were arranged inappropriately in the refrigerators and were mixed with other items including expired and partially used vaccine vials. (Vogel, et al, 2011)
In a cross-sectional study that was conducted in Toronto Canada from August to December 1992, staff responsible for vaccine storage were interviewed about their knowledge and practices of vaccine handling and storage. Refrigerators were inspected, fewer than 7 (6%) practices staff answered all questions related to vaccines storage and handling correctly, and only 11 (10%) refrigerator had a thermometer.
One –third of refrigerators had temperatures outside the recommended range of 2 to 8 degrees centigrade. Older refrigerators were more likely to had inappropriate temperature than newer ones. Knowledge and practice of vaccine storage and handling were often inadequate in primary care physician’s offices (Yuan, et al, 1995)… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
METHODOLOGY
Study Area and Population
The study was conducted in Delta state. Delta state is estimated to have 203 public health facilities (MOHSW, 2010). The study units were community pharmacies and the study populations were people selected for an interview at the community pharmacies in the region.
Study Design
It was a cross-sectional survey. Staff responsible for vaccine availability and management was interviewed about their knowledge and practices of vaccine availability, handling, and storage. Refrigerators and freezers were inspected and temperature documented from freezer tags and observation of vaccine storage conditions… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
RESEARCH FINDINGS AND DISCUSSION
This chapter provides a detailed analysis of the data collected from the field. It starts by analyzing data according to the research questions which guided this study. Various nonparametric tests were used at various stages of data analysis.
Response Rate
Of the selected 40 public health facilities and 5 warehouses (i.e. district hospitals, health centers, and dispensaries). All 40 public health facilities and 5 warehouses respondents agreed and participated in the study. Of the selected 90 people for interviewees only 84 contacted, missing was due to inadequate health workers at facility level working at vaccination area. The overall response rate of the interview that was conducted is 94.7%, however, minor discrepancies in some respondents were observed due to respondents’ inability to provide answers to some of the questions caused by lack of training and less experienced in the vaccination area. Those cases were considered as missing values in the analysis.
Availability of vaccines at the selected community pharmacies
This was done during visiting the facility in the survey and the availability of six key vaccines used in routine Immunization was used as tracer vaccines. The availability of vaccines from 40 contacted community pharmacies and 5 vaccine stores were summarized in the tables below
Table 1: Availability of vaccines at Community pharmacies level on a day of visiting N=40
| TRACER VACCINES | Facilities with Vaccines in Percentage (%) |
| BCG VACCINE | 95 |
| PENTAVALENT VACCINE | 95 |
| OPV | 95 |
| MEASLES VACCINES | 95 |
| TETANUS TOXOID VACCINE | 97.5 |
| RABIES VACCINE | 15 |
| Mean(Average) | 82 |
From table 1: above the least available vaccine at community pharmacies was Rabies vaccine (15%; n=40). Mean availability of vaccines at community pharmacies level is 82% & median is 95%… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
Average Stock out Duration at the Community Pharmacies
Average stock out duration of six tracer vaccines was determined as a means of finding out if vaccines were available at all levels of community pharmacies for the past ten months. The number of days out of stock was calculated within ten months retrospectively.
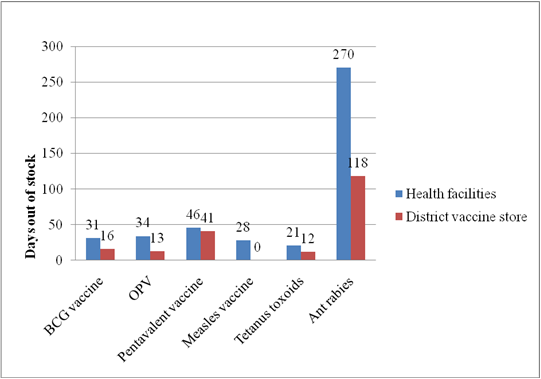
Figure 4: Average stock-out days of vaccines between community pharmacies and district vaccine stores
The study shows that the average stock out days of vaccines between community pharmacies and district… (Scroll down for the link to get the Complete Chapter One to Five Project Material)
CONCLUSIONS AND RECOMMENDATIONS
Conclusions
Managing availability system at public health facilities is highly associated with many challenges that cannot be sorted out by just one player but rather all players involved in the vaccines availability system such as MSD, Regional Vaccine Store, District Vaccine Store, district councils authorities and the Ministry of Health and Social Welfare at large.
The study has identified about 8 main challenges that hinder effective availability system of vaccine; these include unreliable/inadequate vehicles for the availability of vaccines to public community pharmacies, availability from a regional level to the district level and district level to community pharmacies. Stockout condition the study reveals average stock out days was high at the lower health facility level as compared to the…. (Scroll down for the link to get the Complete Chapter One to Five Project Material)
Recommendations
The following are recommendation in response to the above challenges revealed by the study:
- MOHSW/EPI –Need to involve pharmaceutical personnel in the supply chain of vaccines because the study reveals no involvement of pharmaceutical personnel in this programme.
- Pharmacists are custodian in medicines once involved in the programme it’s our hope the problems of poor storage practices, poor quantification of need and storing vaccines out of recommended temperature range can be reduced and controlled under his/ her supervision… (Scroll down for the link to get the Complete Chapter One to Five Project Material)

REFERENCES:
Bonhoeffer, J., Heinnger, U., (2007). ―Adverse events following immunization perception and evidence‖ curr opin infect Dis 20(3): 237-46.
Brudon, P., Rainhorn, J.D., Reich, M., (1994). Indicators for monitoring national drug policies.
Euro Health Group, (2007). Nigeria Drug Tracking Study.
Grobowsky, M., et al, (2005). Ghana and Zambia: Achieving equity in availability of Insecticide Treated Bed nets through links with measles vaccination campaign.
Immune Australia website http:// Immunise.health gov.au/vaccine storage html
Indonesia MOA (2011). Cold chain and Vaccine management Assessment for Animal vaccination program in Bali Province.
Management Sciences for Health (MSH), 1997: Managing Drug Supply, 2nd Edition, Kumarian Press, Connecticut, USA. Pg 317-20,332,347-48,366-68.
MSH, (1995). Rapid pharmaceutical assessment — An indicator-based approach. Washington DC: MSH.
Meri, S., Jorden, M., Jarva, H., (December 2008). ―Microbial complement inhibitors as vaccines‖ vaccine 26 suppl 8, 1113-7.
MOHSW (2008). Immunization coverage survey.
MOHSW (2008). In-depth Assessment of the Medicines Supply System in Nigeria Report).
MOHSW (2010). Nigeria Mainland EPI Review Pg 5, 16, 22.
MOHSW (2009). Vaccine Management Assessments.
National Vaccine Storage guidelines http//www.ag.gov.au/cca.
Stern, A.M., Markel, H., (2005). The history of vaccines & immunization: Health Aff. 24(3) 611-21
Strive FOR five, (2005).National Vaccines Storage guidelines. Retrieved April 2005, from http//www.ag.gvr au/cca.
Subramanyam, K., et al (1989). Vaccine Availability: an operations research study.
Van Sant J.E., (2008). ―The vaccinator: smallpox, medical knowledge, and the opening of Japan‖ JHist Med Allied Sci, 62(2) 276-9.
WHO, 1997. Comparative analysis of national drug policies. Report of second workshop. Geneva: DAP/97.6.
WHO Report, (2008). Global pandemic influenza action plan to increase vaccine supply. Retrieved April 2009, from www.who.int/vaccine documents/
WHO (2008), Health Metrics Network, http/www.healthmetricsnetwork.org.
WHO (2005), Vaccine management assessment. Retrieved April 2005, from www.who.int/ Vaccine-documents http//www.ag.gvr.au/cca
Yuan, L., et al (July 1995).Vaccine storage and handling. Knowledge and practice in primary care physicians’ officers. 41, 1169-76.
(Scroll down for the link to get the Complete Chapter One to Five Project Material)

(Get the Complete Chapter One to Five Project Material)
| MESSAGE US Need quick, reliable writing support? Message us Now and we’ll match you with a professional writer who gets results! or email your files to [email protected] |