EFFECTS OF VITAMIN SUPPLEMENTATION ON LEAD TOXICITY IN WISTAR ALBINO RATS
ABSTRACT
Effects of vitamin supplementation on lead toxicity in Wistar Albino rats over a short term (3 weeks) were investigated. Studies have revealed that lead has a wide range of health effects that can result from exposure, and that lead can cause health effects at blood lead levels previously thought to be safe. An increasing body of evidence suggests that lead is associated with a number of health conditions.
Twenty Wistar albino rats were randomly divided into four experimental groups of five rats each, Control group were fed normal rat feed with distilled water, Group 2,3 and 4 were fed normal rat feed, water and received daily oral administration of lead acetate 250mg/kg daily. In addition, groups 3 and 4 received 100mg/kg and 150mg/kg oral administration of vitamin C respectively, for three weeks.
The mild effect of lead acetate was observed in hematological parameters as indicated by a slight increase, which was not statistically significant, in RBC and MPV… (Scroll down for the link to get the Complete Project Material)

INTRODUCTION
Lead (Pb) toxicity is probably the most common form of heavy metal intoxication. It is well-documented as one of the most dangerous and insidious poisons. Its continuous environmental and occupational exposure may contribute to renal, nervous, hepatic, hematological, and reproductive disorders in man and animals (Flora et al., 2006; El-Sayed and El-Neweshy, 2009, Ashry et al., 2010).
The absorbed Pb is conjugated in the liver and passed to the kidney, where a small quantity is excreted in urine and the rest accumulates in various body organs and affects many biological activities at the molecular, cellular and intercellular levels, which may result in morphological alterations that can remain even after Pb levels have fallen (Jarrar, 2003; Sidhu and Nehru, 2004; Taib et al., 2004; Flora et al., 2006). Lead-induced oxidative stress or disruption of prooxidant/antioxidant balance in the blood and other soft tissues has been postulated to be the major mechanism of Pb associated tissue injury (Pande et al., 2001; Flora et al., 2003).
It causes oxidative stress by inducing the generation of reactive oxygen species (ROS) (Gurer and Ercal, 2000), increasing the level of lipid peroxidation and thiobarbituric acid-reactive substances (Adonaylo and Oteiza, 1999; Upasani et al., 2001; Flora et al., 2003; Ashry et al., 2010), and inhibiting the activity of many antioxidant enzymes, including glutathione (Sidhu and Nehru, 2004; Flora et al., 2006; Ashry et al., 2010). Consequently, Pb alters the antioxidant defense system of cells resulting in pathophysiological events in various body organs.
Hence, the therapeutic strategy of strengthening the cell’s antioxidant capacity may fortify the longterm effective treatment of Pb poisoning. This may be achieved through exogenous supplementation of antioxidant molecules as an alternative to chelation therapy (Gurer and Ercal, 2000; Flora et al., 2003; Ashry et al., 2010)… (Scroll down for the link to get the Complete Project Material)
Statement of the problem
Lead ubiquitous environmental toxicants with high exposure potential to man (Hughes et al., 2011; Mankidy et al., 2013). They are known to induce oxidative stress (Jomova et al., 2011; Erkekoglu et al., 2010) and are reported to be endocrine disruptors (Davey et al., 2007; Li et al., 2012). Both environmental agents are found in the air, soil, drinking water, and food (Du et al., 2010; Rudel et al., 2003; Adeyemi et al., 2009; Kamrin, 2009)… (Scroll down for the link to get the Complete Project Material)
Research Objective
This study aimed to investigate the ameliorative effect of VC against chronic Pb-associated direct tissue damage in the liver, kidneys, brain, and testes of male rats by observing histopathological alterations… (Scroll down for the link to get the Complete Project Material)
LITERATURE REVIEW
Conceptual Review
Lead is a widely used metal, but it is simultaneously a versatile, subtle, and persistent poison. Significant exposure to lead is an environmental threat to optimal health and physical development in young children that affects all socioeconomic groups [1]. However, the deleterious effects of lead may be efficiently prevented by applying specific regulations to its use. Metallic lead has constituted a part of the human environment for over 5000 years [2].
The characteristic features of lead toxicity, including anemia, colic, neuropathy, nephropathy, sterility, and coma, were noted both by Hippocrates and Nikander in ancient times [3]. In 370 BC, Hippocrates first described abdominal colic in a man who mined metals [2]. The effects of lead toxicity on young children were first described in 1892 in Brisbane, Australia [4].
Even though two thousand years have passed since Vitruvius chronicled the dangers of lead in water supplies, this threat to public health still remains with us [5]… (Scroll down for the link to get the Complete Project Material)
- Absorption of Lead
Lead may enter the body by ingestion through the intestines, through the lungs by inhalation, through the skin, or by direct swallowing and ingestion [9]. Inorganic lead absorption takes place throughout the respiratory and gastrointestinal tracts. For adults with occupational exposure, the most significant route for absorption is through the respiratory tract [2,9]. Respiratory lead absorption is primarily dependent on particle size.
The percentage of inhaled lead reaching the bloodstream is estimated to be 30–40% [2]. Rates of absorption through the gastrointestinal tract depend on the nutritional status and the age of the individual exposed. Therefore, while adults absorb an average of 10 to 15% of the ingested quantity, this amount can increase to 50% in infants, young children, and pregnant women [2,9]… (Scroll down for the link to get the Complete Project Material)
RESEARCH METHODOLOGY
Materials and Methods
- Chemicals
Lead-acetate [(C2H3O2)2Pb 3H2O] (PbAc) and sodium-acetate [CH3COONa 3H2O] (NaAc) were obtained from Sigma Chemical Co. (St. Louis, MO). Vitamin C (ascorbic acid) was obtained from Pharco Pharmaceutical Company, Alexandria, Egypt. All other chemicals and reagents used were of analytical reagent grade.
- Heavy Metals and Vitamin Preparation
A 0.5% Pb solution was prepared by dissolving 5 g of PbAc in 1000 mL of distilled acidified water to prevent the formation of Pb precipitate. A corresponding solution of NaAc containing the equivalent amount of acetate was prepared and used as a vehicle for the control. Vitamin C dissolved in saline and used freshly in experimental animals… (Scroll down for the link to get the Complete Project Material)
RESEARCH FINDINGS AND DISCUSSION
Hematoxylin and eosin-stained sections of the liver, kidneys, brain, and testes were evaluated under light microscopy. The number of rats with certain types of histopathological findings and their occurrence in PbAc-intoxicated and PbAc-intoxicated VC-supplemented groups are summarized in Table 1.
- Kidneys
The kidneys of control rats exhibited normal renal tissue, where normal glomeruli, tubular epithelium, and interstitial tissue were observed (Fig. 2A). Lead-induced nephropathy was apparent in the PbAc-intoxicated group. Karyomegaly with eosinophilic intranuclear inclusions was abundant (410 per HPF) in the epithelial cells of proximal tubules of six rats and moderate (5–10 per HPF) in another four rats. Glomerular damage and tubular necrosis with invading inflammatory cells were also characteristic lesions (Figs. 2B1 and 2B2).
Rats treated with PbAc plus VC exhibited mild karyomegaly with eosinophilic intranuclear inclusions in the epithelial cells of proximal tubules of five rats and the other five rats were normal. Focal cloudy swelling of the tubular epithelium with normal glomeruli was noted (Fig. 2C).
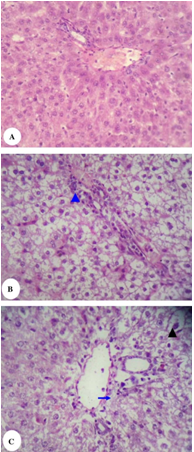
Fig. 1. Photomicrograph of rat liver stained with hematoxylin and eosin… (Scroll down for the link to get the Complete Project Material)
CONCLUSION AND RECOMMENDATIONS
Conclusion
This study evaluates the efficacy of vitamin C (VC) in ameliorating the detrimental effects of long-term lead intoxication on the liver, kidneys, brain, and testes as assessed by histopathology. A total of forty male Wistar rats (six-weeks-old) was divided into 4 groups: control group; lead-acetate (PbAc)-treated group (20 mg PbAc/kg bwt); PbAc+VC-treated group (20 mg PbAc/kg bwt plus 20 mg VC/kg bwt); and VC-treated group (20 mg VC/kg bwt).
The experimental period lasted for 60 successive days in which PbAc was administered once daily while VC was supplemented every other day using intragastric intubation. At the end of the experimental period, all rats were sacrificed and pathological examinations were performed. Control and VC-supplemented rats showed normal liver, kidney, brain, and testes histology… (Scroll down for the link to get the Complete Project Material)

REFERENCES:
Centers for Disease Control and Prevention. Preventing Lead Poisoning in Young Children: A Statement by the Centers for Disease Control, October 1991. Atlanta, GA: US Dept of Health and Human Services, 1991
Philip AT, Gerson B. Lead poisoning – Part I. Clin Lab Med, 1994; 14: 423–44
Landrigan PJ, Silbergeld EK, Froines JR, Pfeffer RM: Lead in the modern workplace. Am J Public Health, 1990; 80: 907–8
Fison DC: The Royal Children’s Hospital, Bisbane, 1878 to 1978. Med J Aust, 1978; 2: 137–38
Russell Jones R: The continuing hazard of lead in drinking wa-ter. Lancet, 1989; 2: 669–70
Needleman HL: Childhood lead poisoning: the promise and abandonment of primary prevention. Am J Public Health, 1998; 88: 1871–77
Rosner D, Markowitz G: A “gift of God”?: The public health con-troversy over leaded gasoline during the 1920s. Am J Public Health, 1985; 75: 344–52. (Wistar Albino Rats)
Needleman HL: Clamped in a straitjacket: the insertion of lead into gasoline. Environ Res, 1997; 74: 95–103. (Wistar Albino Rats)
Markowitz M: Lead Poisoning. Pediatr Rev, 2000; 21: 327–35. (Wistar Albino Rats)
Rabinowitz MB, Kopple JD, Wetheril GW: Effects of food intake and fasting on gastrointestinal lead absorption in humans. Am J Clin Nutr, 1980; 33: 1784–88. (Wistar Albino Rats)
Fischbein A: Occupational and environmental exposure to lead. In: Environmental and occupational medicine. Rom, WN (Ed), Philadelphia, Lippincott-Raven Publishers, 1998; 973. (Wistar Albino Rats)
Rabinowitz MB: Toxicokinetics of bone lead. Environ Health Perspect, 1991; 91: 33–37. (Wistar Albino Rats)
Skerfving S, Nilsson U, Schutz A, Gerhardsson L: Biological monitoring of inorganic lead. Scand J work Environ. Health, 1993; 19(Suppl.1): 59–64. (Wistar Albino Rats)
Roberts JR, Reigart JR, Ebeling M, Hulsey TC: Time required for blood lead levels to decline in nonchelated children. J Toxicol Clin Toxicol, 2001; 39: 153–60. (Wistar Albino Rats)
Guidelines for drinking water quality, 2nd ed. Vol. 2. Health criteria and other supporting information. Geneva, World Health Organization, 1996; 254–75. (Wistar Albino Rats)
Rabinowitz MB, Wetherill CW, Kopple JD: Kinetic analysis of lead metabolism in healthy humans. J Clin Invest, 1976; 58: 260–70. (Wistar Albino Rats)
Silbergeld EK: Lead in bone: implications for toxicology during pregnancy and lactation. Environ Health Perspect, 1991; 91: 63–70. (Wistar Albino Rats)
Gulson BL, Mahaffey KR, Jameson CW et al: Mobilization of lead from the skeleton during the postnatal period is larger than during pregnancy. J Lab Clin Med, 1998; 131: 324–29. (Wistar Albino Rats)
Klein M, Barbe F, Pascal V et al: Lead poisoning secondary to hyperthy-roidism: report of two cases. Eur J Endocrinol, 1998; 138: 185–88. (Wistar Albino Rats)
Tothill P, Matheson LM, Mckay K, Smyth JF: Mobilisation of lead by cis-platin. Lancet, 1989; 2: 1342. (Wistar Albino Rats)
Silbergeld EK, Schwartz J, Mahaffey K: Lead and osteoporosis: mobilization of lead from bone in postmenopausal women. Environ Res, 1988; 47: 79–94. (Wistar Albino Rats)
NRC, National Research Council. Measuring lead exposure in infants, children, and other sensitive populations. Washington, D.C.: National Academy Press, 1993. (Wistar Albino Rats)
Mortada WI, Sobh MA, El-Defrawy MM: The exposure to cadmium, lead and mercury from smoking and its impact on renal integrity. Med Sci Monit, 2004; 10(3): CR112–CR116. (Wistar Albino Rats)
Shannon MW, Graef JW: Lead intoxication in infancy. Pediatrics, 1992; 89: 87–90. (Wistar Albino Rats)
Woolf DA: Aetiology of acute lead encephalopathy in Omani infants. J Trop Pediatr, 1990; 36: 328–30. (Wistar Albino Rats)
Abdullah MA: Lead poisoning among children in Saudi Arabia. J Trop Med Hyg, 1984; 87: 67 – 70. (Wistar Albino Rats)
Shaltout A, Yaish SA, Fernando N: Lead encephalopathy in infants in Kuwait. A study of 20 infants with particular reference to clinical presentation and source of lead poisoning. Ann Trop Paediatr, 1981; 1: 209–15. (Wistar Albino Rats)
(Scroll down for the link to get the Complete Project Material)

(Get the Complete Project Material)
| MESSAGE US Need quick, reliable writing support? Message us Now and we’ll match you with a professional writer who gets results! or email your files to [email protected] |




